Cancer is a very complex and challenging disease to treat, which is why the medical community is always searching for more effective treatment methods with fewer negative side effects.
One of the most critical steps in developing new forms of treatment and improving patient outcomes is through clinical trials. These trials offer hope to patients by giving them access to cutting-edge treatments that may be more effective.
The Cancer and Hematology Centers (CHC) has partnered with START Midwest to offer dozens of clinical trials to our patients. There are cancer clinical trials available for multiple types of cancer, including breast, lung, and gastrointestinal cancers.
If you’re interested in joining a clinical trial, your care team can determine if you are eligible and talk with you more about the process. Call us today at (616) 975-3065 to get started.
What Is a Clinical Trial for Cancer?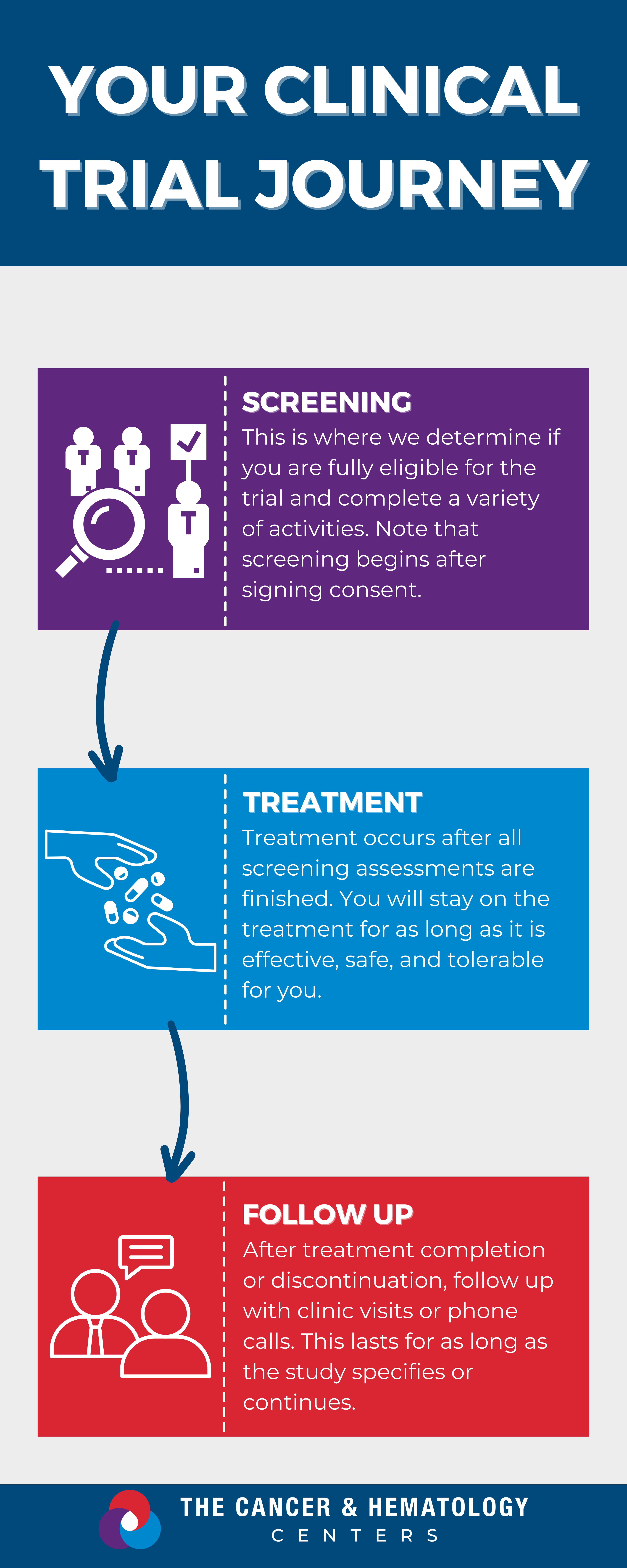
Cancer clinical trials try to find new medications and treatments to improve the cure of cancer. They may also seek to minimize the unwanted side effects of treatment.
Patients who join cancer clinical trials have the opportunity to receive forms of cancer treatment that may not be widely available for many more years. These cutting-edge methods may prove more effective than existing treatments.
By the time a new medication reaches the clinical trial phase, it will have gone through years of research to understand its effects on cancer cells. Tests will have been performed in laboratories on cancerous tissue and on animals. Before you join a trial, the researchers and your care team of doctors, nurses, nutritionists, and social workers will talk with you about the steps and precautions that have been taken to test its safety and efficacy and how it may benefit you.
Who Is Eligible for Cancer Clinical Trials?
There is a common belief that only people with late-stage or terminal cancer are able to participate in clinical trials, but that is not the case; patients of any cancer stage can join a trial—and often, the earlier, the better.
Each clinical trial is designed specifically for certain people with certain cancers. Eligibility is based on many factors, such as the treatment being tested, the phase of the trial, your cancer stage, and medical condition.
How Successful Are Clinical Trials for Cancer?
Clinical trials are the key to making progress against cancer. By looking closely at all your treatment options, including clinical trials, you are taking an active role in a decision that affects your life. The success of a clinical trial depends on the clinical trial phase, your cancer and your personal goals. Your care team will work with you to personalize your treatment plan to ensure success however you define it.
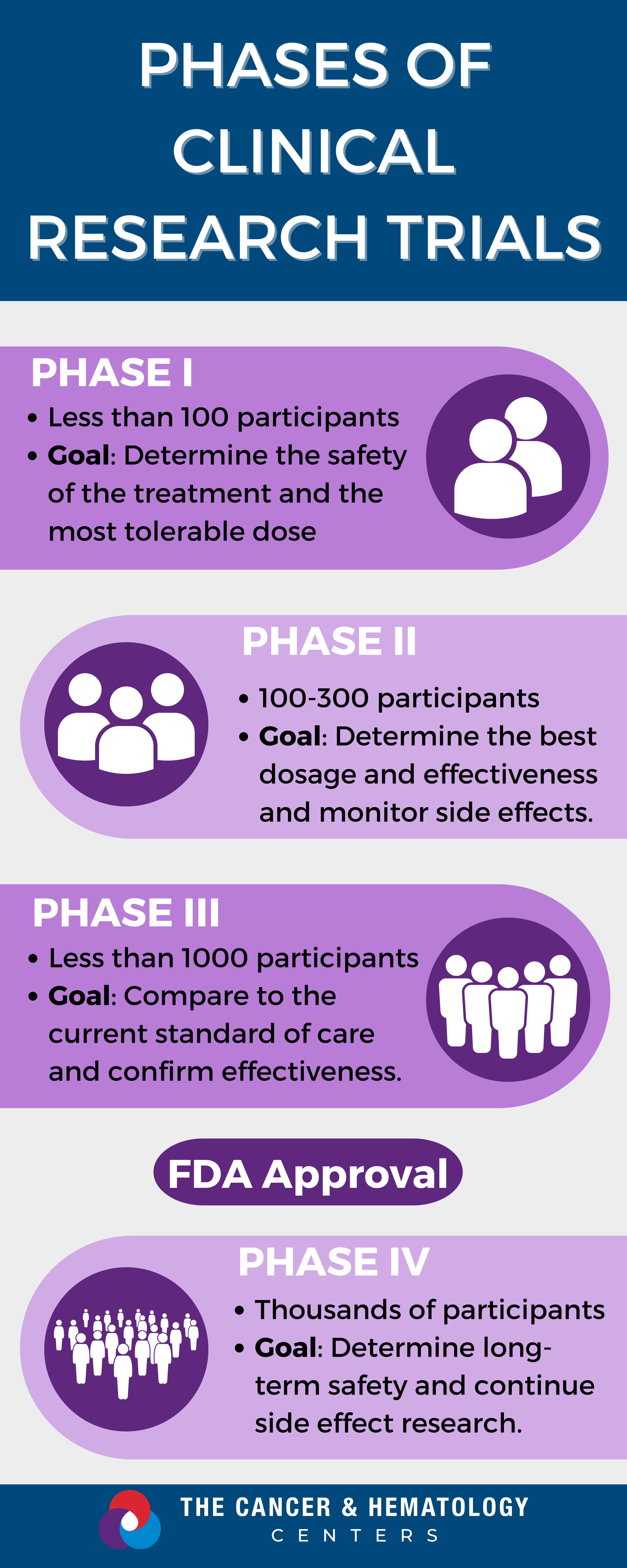
Cancer Clinical Trial Phases
Clinical trials are completed in four distinct phases, and The Cancer and Hematology Centers participates in each of them with our partner, START Midwest, making us part of the world’s largest Phase I clinical trial network.
Phase I Trials
The first phase of cancer clinical trials seeks to evaluate whether a treatment is safe and what the proper dosage should be. Phase I trials also look for potential side effects of the treatment. They typically include a small number of patients, around 20–80, who have not significantly benefited from existing treatment methods.
Phase II Trials
In the second phase, researchers apply their findings of medication doses and treatment schedules to a new group of patients. The number of patients involved in the trial is increased for the second phase.
Phase III Trials
During Phase III, the number of patients is increased to hundreds or thousands, and the best known treatment is compared to what may be even better.
Phase IV Trials
The last phase of a cancer clinical trial involves a much larger group of patients and collects any additional side effects that may come up with treatment over a long period of time.
Clinical Cancer Trials at CHC
Choosing whether to join a clinical trial is a major decision in your treatment and should be made with expert advice and a wealth of information. Our care team would be happy to talk with you in depth about the process of joining a trial and which options are best for you. It’s important that you feel comfortable that you are making the right decision for your health.
The trials we offer are overseen by the Food and Drug Administration (FDA), an institutional review board, and a physician who is specially trained to monitor clinical trials and ensure patient safety.
Our START Midwest Partnership
CHC has partnered with START Midwest because they are a leader in cancer research and have had led the way on the use of many oncology medications that are approved today. Our relationship enables CHC patients to access the largest Phase I trial research network in the world.
Our Current Clinical Cancer Trials
There are dozens of trials currently available at CHC. If none of the trials meet your needs, there are more offered through the National Institutes of Health (NIH).
Call Today About Cancer Clinical Trials at CHC
If you’re interested in receiving cancer care at CHC or participating in a cancer clinical trial give us a call at (616) 975-3065. Our staff can set up an appointment and help you determine your eligibility.
Frequently Asked Questions
Are Cancer Clinical Trials Free?
Trials aren’t free, but some costs may be covered by your insurance company. Our financial coordinators can talk with you about your coverage and financial options.
Are Placebos Used in Cancer Clinical Trials?
A placebo is a treatment that does not have an active drug and is used in some clinical trials to ensure that the benefits of a drug are from the ingredients and not the psychological effect of taking a drug. When participating in a clinical trial, the researcher will tell you if you have a chance of getting a placebo.
What Percent of Cancer Patients Participate in Clinical Trials?
It is estimated that less than 10% of adult cancer patients enroll in clinical trials. Patients may not enroll because they are already receiving effective treatment, are worried about the risks, or aren’t eligible.